GSK announces new Shingrix data demonstrate 100% vaccine efficacy in the prevention of shingles in adults aged 50 and over in China
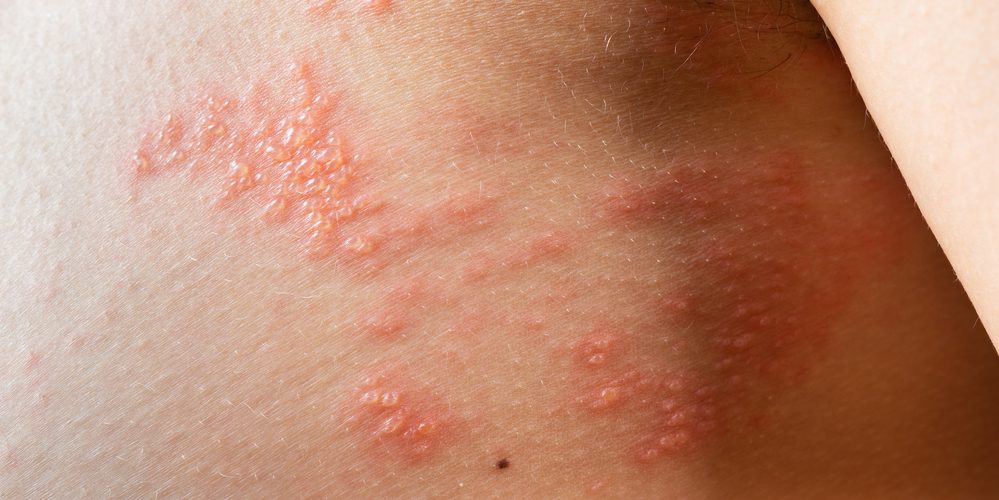
GSK plc announced positive results from the first-ever efficacy trial of Shingrix (Recombinant Zoster Vaccine or RZV) in China. These results come from the post-license phase IV trial (ZOSTER-076), which evaluated the efficacy and safety of RZV in preventing shingles in adults aged 50 and over. The trial included almost 6,000 participants randomised 1:1 to the RZV or placebo group and followed in an observer-blind design. No cases of shingles were reported among the participants who received RZV, compared to 31 cases in the placebo arm.
The results are in line with findings from the pivotal phase III trials ZOE-50 and ZOE-70 investigating the efficacy and safety of RZV, showing vaccine efficacy was up to 97%2 in adults aged 50 and over, over a follow-up period of approximately four years. The safety profile observed in this trial was consistent with the established safety profile of the vaccine. The new data add to the growing body of evidence affirming the efficacy and safety profile of RZV in preventing shingles in adults aged 50 and over.
Globally, the varicella zoster virus (VZV) is present in over 90% of adults. VZV remains dormant in the nervous system, waiting to reactivate as shingles with advancing age. The population of people aged 65 and over in China is rapidly increasing, with a predicted percentage increase from 6.8% in 2000 to 23.6% by 2050. It is estimated that there are approximately 6 million cases of shingles in China each year, and this number is expected to increase in line with local population ageing.