FDA approves redosable gene therapy to treat DEB
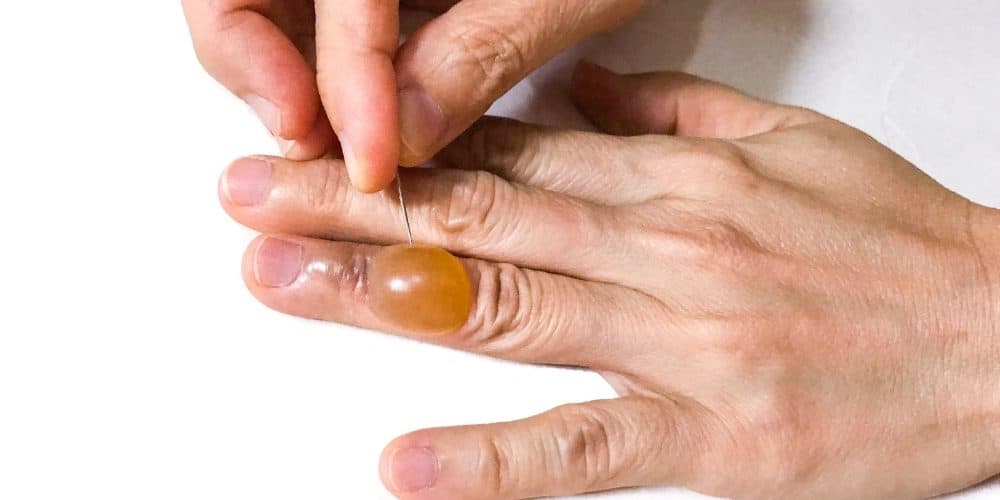
Krystal Biotech, Inc., a biotechnology company focused on developing and commercializing genetic medicines for patients with rare diseases, announced the US Food and Drug Administration (FDA) has approved Vyjuvek (beremagene geperpavec-svdt) for the treatment of patients six months of age or older with dystrophic epidermolysis bullosa (DEB).
Vyjuvek is designed to address the genetic root cause of DEB by delivering functional copies of the human COL7A1 gene to provide wound healing and sustained functional COL7 protein expression with redosing. Vyjuvek is the first-ever redosable gene therapy and the first and only medicine approved by the FDA for the treatment of DEB, both recessive and dominant, that can be administered by a healthcare professional in either a healthcare professional setting or in the home.
DEB is a rare and serious disease that affects the skin and mucosal tissues caused by one or more mutations in the COL7A1 gene. The COL7A1 gene is responsible for the production of functional COL7 protein that forms anchoring fibrils necessary to bind the dermis (inner layer of the skin) to the epidermis (outer layer of the skin). The lack of functional anchoring fibrils in DEB patients leads to extremely fragile skin that blisters and tears with minor friction or trauma. DEB patients suffer from open wounds, which lead to recurrent skin infections and fibrosis that can cause fusion of fingers and toes, and ultimately increase the risk of developing an aggressive form of skin cancer.